Cancer Stem Cells as Potential Targets of Phytotoxic Alkaloids from the Toxic Plants-Phytotoxins Database
Main Article Content
Abstract
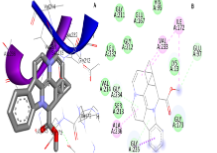
Targeting cancer stem cells (CSCs) in anticancer discovery is very difficult due to the resistance of CSCs to conventional drugs. Different CSC targets, such as the ABC cassette, surface markers, signal cascade, and tumour microenvironment, are involved in the interruption of cell signalling pathways that are critical for the survival and functioning of the CSC population. The study aimed to identify potential drug-like phytotoxic alkaloids with anticancer activity from the toxic plants-phytotoxins (TPPTs) database. A total of 1586 phytotoxins were filtered to obtain 653 alkaloids. Lipinski’s properties and the TPSA were predicted for drug likeness and toxicity based on various organ endpoints. Compounds that obeyed Lipinski’s rule of five, with moderate or no toxicity were selected. The 11 drug-like phytotoxic alkaloids obtained from the filtering were docked on an isomerase-perdeuterated E65Q-TIM protein (ID: 7AZA; resolution = 1.10 Å) co-crystallized with phosphoglycolohydroxamate. The best binding poses were ranked using their binding energies (E) and inhibition constants (Ki). An evaluation of the protein—ligand’s best conformational poses allowed us to identify three indole alkaloids (catharanthine, (-)-eburnamonine and apovincamine) with significant protein—ligand binding interactions for (-)-eburnamonine (E = −8.03 kcal/mol; Ki = 1.30 μM), catharanthine (E = −8.229 kcal/mol; Ki = 0.910 μM) and apovincamine (E = −8.40 kcal/mol; Ki = 0.701 μM). All the docked ligands could bind more efficiently to the target than phosphoglycolohydroxamate. The efficient inhibition of perdeuterated E65Q-TIM in CSCs using phytotoxic alkaloids provided more insights into understanding the mechanisms of the anticancer activity of phytotoxic alkaloids.
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
References
Ayoka TO, Nwachukwu N, Ene AC, Igwe CU, Nnadi CO. The hepatocurative effects of Zanthoxylum zanthoxyloides alkaloids on tetrachloromethane-induced hepatotoxicity on albino rats. Indian J Clin Biochem. 2024; 39(2): 186-197.
Ayoka TO, Ngwu N, Ene AC, Igwe CU, Nnadi CO. Liquid chromatography-mass spectrometric analyses of potential antioxidant constituents from Zanthoxylum zanthoxyloides leaves: Probing into the role of alkaloids. Trop J Nat Prod Res. 2020; 4(10): 817-823.
Ayoka TO, Nwachukwu N, Ene AC, Igwe CU, Nnadi CO. Hepatocurative and histopathological evaluations in albino rats exposed to Vitex doniana alkaloids. Lett Applied NanoBioSci. 2023; 12(2): 56.
Ayoka TO, Ezema BO, Ani NI, Nnadi CO. The role of alkaloids in the management of cardiovascular disorders. In: Natural Products: An answer to cardiovascular complications. Upaganlawar AB and Joshi T (Eds). 2023, Nova Science Publishers, Inc. pp 57-74.
Olofinsan K, Abrahamse H, George BP. Therapeutic roles of alkaloids and alkaloid derivatives in cancer management. Molecules. 2023; 28(14): 5578.
Ouverney G, Hottz D, Robbs BK. Drug resistance and novel targets for cancer therapy: An overview of recent findings. Biomed. 2024; 12(4): 816.
Tilsed CM, Fisher SA, Nowak AK, Lake RA, Lesterhuis WJ. Cancer chemotherapy: Insights into cellular and tumour microenvironmental mechanisms of action. Front. Oncol. 2022, 12,960317.
Udeani TK, Ugwu LO, Nnadi CO, Okwuosa CN. Evaluation of anti-microbial activity and proximate composition of alkaloids from Vitex doniana seed. Dhaka Univ J Pharm Sci. 2021; 20(1): 81-86.
Ye J, Wu J, Liu B. Therapeutic strategies of dual-target small molecules to overcome drug resistance in cancer therapy. Biochim Biophys Acta. 2023; 1878(3): 188866.
Lathia J, Liu H, Matei D. The clinical impact of cancer stem cells. The Oncologist. 2020; 25(2): 123-131.
Liang L, Kaufmann AM. The significance of cancer stem cells and epithelial-mesenchymal transition in metastasis and anticancer therapy. Int J Molec Sci. 2023; 24(3): 2555.
Lee TKW, Guan XY, Ma S. Cancer stem cells in hepatocellular carcinoma-From origin to clinical implications. Nature Rev Gasroenterol Hepatol. 2022; 19(1): 26-44.
Ajmeera D, Ajumeera R. Drug repurposing: A novel strategy to target cancer cells and therapeutic resistance. Genes Dis. 2024; 11(1): 148-175.
Skvortsorva I. Cancer stem cells: What do we know about them? Cells. 2021; 10(6): 1528.
Günthardt BF, Hollender J, Hungerbühler K, Scheringer M, Bucheli TD. Comprehensive Toxic Plants-Phytotoxins database and its application in assessing aquatic micro pollution potential. J Agric Food Chem. 2018; 66(29): 7577–7588.
Daina A, Michielin O, Zoete V. SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci Rep. 2017; 7(1): 42717.
Banerjee P, Eckert AO, Schrey AK, Preissner R. ProTox-II: A webserver for the prediction of toxicity of chemicals, Nucleic Acids Res. 2018; 46(2018): W257–W263.
Kelpsas V, Caldararu O, Blakeley MP, Coquelle N, Wierenga RK, Ryde U, von Wachenfeldt C, Oksanen E. Neutron structures of Leishmania mexicana triosephosphate isomerase in complex with reaction-intermediate mimics shed light on the proton-shuttling steps. IUCrJ. 2021; 8(4): 633 – 643.
Nnadi CO, Althaus JB, Nwodo NJ, Schmidt TJ. A 3D-QSAR study on the antitrypanosomal and cytotoxic activities of steroid alkaloids by comparative molecular field analysis. Molecules. 2018; 23: 1113.
Ezema IF, Akwu VC, Didigwu OK, Ogbonna JE, Ugwu AC, Nnadi CO. Exploring different drug targets responsible for the inhibitory activity of N, N′-substituted diamine derivatives in Leishmania. Engineer Proc. 2023; 56(1): 178.
Lipinski CA. Lead- and drug-like compounds: The rule-of-five revolution. Drug Discov Today Technol. 2004; 1(4): 337–341.
Didigwu OK, Nnadi CO. Theoretical drug-likeness, pharmacokinetic and toxicities of phytotoxic terpenes from the Toxic Plants-PhytoToxins. Trop J Nat Prod Res. 2024; 8(10): 8867 – 8873
Nnadi CO, Ozioko LU, Eneje GC, Onah CM, Obonga WO. In-vivo antitrypanosomal effect and in silico prediction of chronic toxicity of N-methylholaphyllamine in rats. Trop J Pharm Res. 2020; 19(11): 2369-2375.
Nnadi CO, Ngwu SO, Ohagwu MBZ. In-vivo and in-silico evidence of antitrypanocidal activities of selected plants from Asteraceae family against Trypanosoma brucei brucei. Bioint Res Appl Chem. 2023; 13(1): 30
Nnadi CO, Banda SH, Uzonwanne MU, Didigwu OK. Cancer Stem Cells as Potential Targets of Phytotoxic Alkaloids: Drug-likeness Prediction and Molecular Docking Studies. The 4th International Electronic Conference on Cancers held on 06-08 March, 2024, Online. https://sciforum.net/paper/view/16913